
0. Abstract
In medical device CNC machining, the MOQ is more than just a pricing factor. It determines whether the manufacturing process can maintain stability beyond the initial batch. While early runs may pass inspection, variations often emerge later if the production window is not clearly defined.
Process stability in implants and surgical instruments relies on validated programs, stable fixturing, controlled tooling, and consistent cutting conditions. This article explains how these factors influence MOQ decisions in medical OEM projects and how teams can mitigate manufacturing risks during scale-up.
Procurement managers evaluating supplier capabilities and R&D teams planning pilot-to-production transitions will find this guide especially relevant to their daily challenges.

Table of Contents
-
Fundamentals: What Does MOQ Mean in Medical Device Machining?
-
Conclusion: A Structured Approach to Balancing Cost and Risk
-
YSF Medical: Your Precision OEM Partner in Medical Device Manufacturing
1. Fundamentals: What Does MOQ Mean in Medical Device Machining?
1.1 The Business Logic Behind MOQ
In medical device CNC machining, the MOQ is not merely about whether a supplier will accept the order. It also determines whether the batch size is sufficient to support a properly controlled production run from the outset.
Before machining begins, teams verify material certifications, assign traceable batch codes, define process documentation, and align inspection plans. These steps require engineering and quality resources regardless of volume, and much of this effort is not reflected in the quote.
When quantities are low, the preparation workload remains constant but is distributed across fewer parts. As production scales, teams often need to rebuild traceability structures and documentation initially designed for small runs, which slows progress and introduces unnecessary complexity.
1.2 Quality Control Costs in Precision Manufacturing
Orthopedic implants and surgical instruments require extremely tight tolerances, sometimes as precise as ±0.01 mm. Before full production begins, teams conduct first article inspections and pilot builds to verify that programs, tooling, fixtures, and measurement systems can consistently reproduce parts. This stage requires significant engineering and inspection effort.
When volumes are too low, the process may never fully stabilize. In subsequent releases, validation steps often restart, increasing variation between runs and leading to additional sampling and schedule delays.
A practical MOQ provides the team with sufficient volume to establish a stable process early on. Without it, variations tend to emerge during scale-up, and initial time savings are often lost due to revalidation.
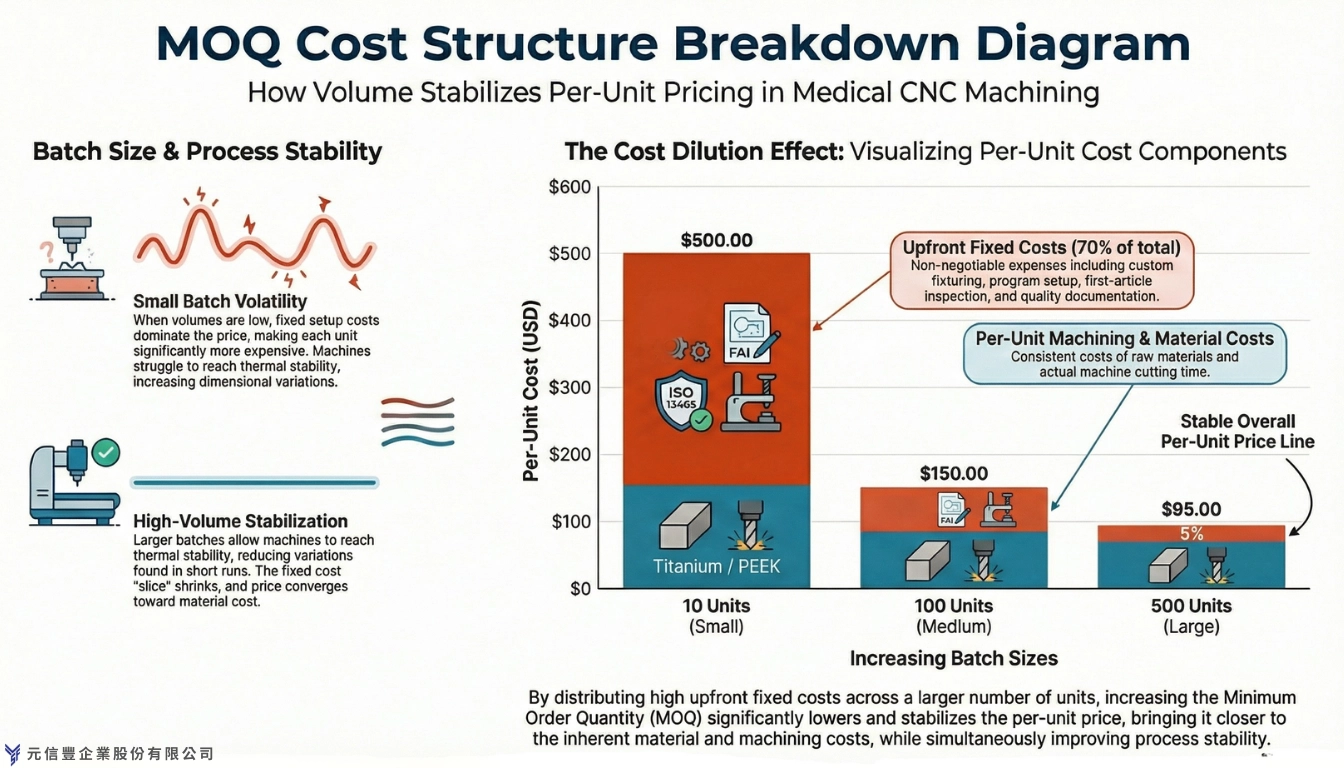
1.3 Batch Size and Process Stability
Many sourcing teams consider MOQ primarily as a pricing term. However, on the shop floor, batch size plays a crucial role in determining whether the machining process can achieve stable operating conditions.
During continuous CNC operations, spindle heat, machine expansion, and coolant flow gradually stabilize. In very short runs, production may conclude before the machine reaches a steady state, potentially causing minor dimensional variations in the initial parts. These variations often become more pronounced as production volume increases, resulting in additional offset adjustments and increased engineering time.
A well-defined MOQ provides the team with sufficient volume to achieve thermal stability and verify that the parameters support sustained production, rather than merely a short batch that passes inspection.
2.MOQ Considerations by Machining Method
2.1 Cost Structure in CNC Milling
Complex medical components, such as bone plates and spinal fixation parts, often require multi-sided CNC milling.Each reorientation necessitates re-fixturing and resetting coordinates, which consumes skilled labor and machine time. These setup steps are essential to meet stringent medical tolerances and traceability requirements.
When batch sizes are small, setup and validation times cannot be distributed across enough units. Increases in unit cost are driven more by preparation and machine occupancy than by the actual cutting time.
Custom fixturing is also essential. Irregular geometries require specialized locating and clamping systems to ensure repeatability. If fixturing is not stabilized early, minor clamping variations can cause dimensional shifts during scale-up, resulting in increased inspection and continuous adjustments.
2-2 The Precision Advantage of Mill-Turn Machining
For implant screws and bone screws, where concentricity and face perpendicularity are critical, mill-turn machining offers a distinct advantage. By integrating turning and milling in a single setup, it preserves geometric relationships and minimizes datum shifts between operations.
When parts move across multiple machines, small reference shifts can accumulate. Although individual dimensions may pass inspection, assembly fit can vary as geometric relationships drift. These issues often arise during scale-up rather than in early samples.
Stabilizing a mill-turn process requires structured programming, precise offset control, and initial first-article trials. Engineers adjust parameters until concentricity and surface finish meet specifications under consistent, repeatable conditions. Establishing a realistic MOQ ensures this work is completed early, allowing production to begin with a validated and stable setup.
3.Hidden Cost Drivers in Medical Device Machining
3-1 Batch Constraints in High-Specification Materials
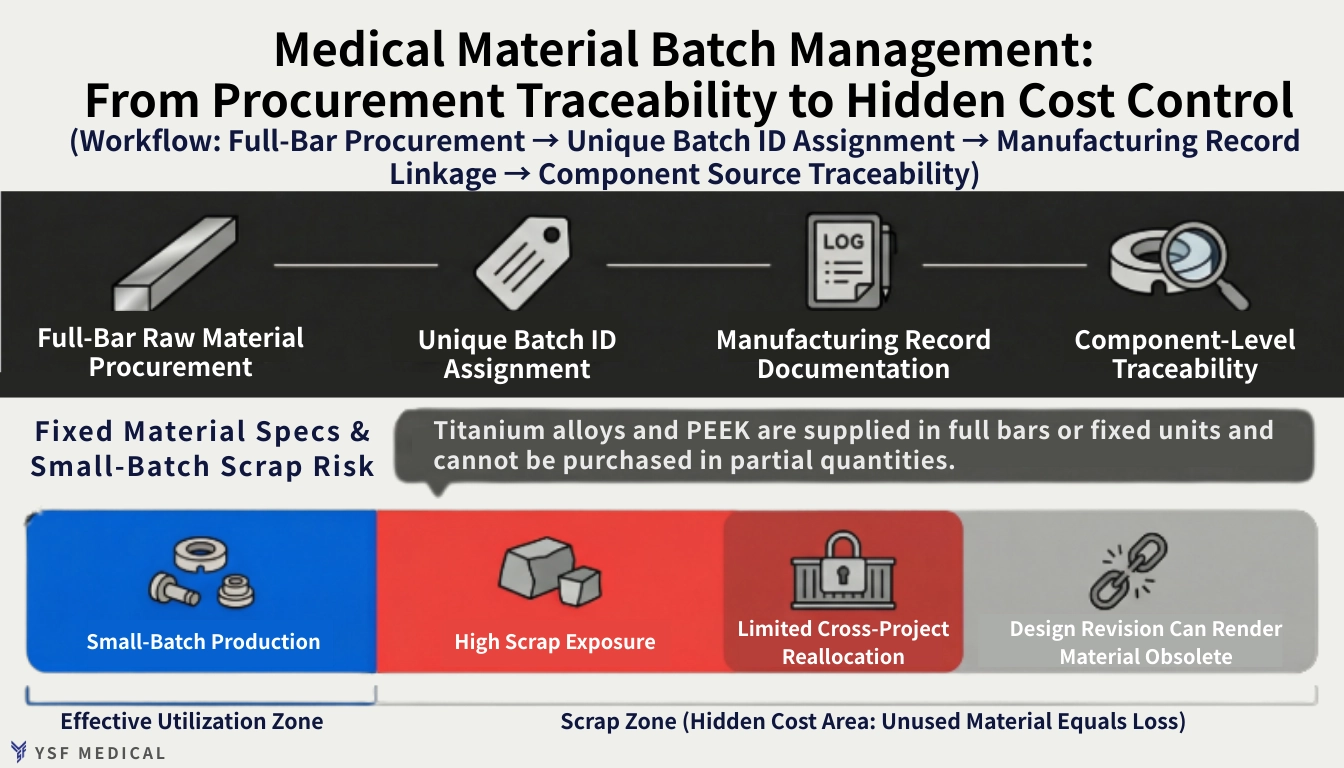
Medical-grade materials such as Ti-6Al-4V ELI and PEEK are typically supplied in standardized forms with minimum order quantities. For example, titanium bar stock is sold in full lengths. Even if only a portion of the bar is used, the manufacturer must manage the remaining material, including storage, identification, and batch control.
Each production lot must correspond to the correct material certification and heat number. Documentation must align with internal quality records to ensure full traceability, regardless of whether the order consists of twenty parts or two hundred.
When volumes are low, the challenge is not material availability but excess inventory. Residual stock may remain unused for months, and design or specification changes can render it obsolete. What seems like a minor purchasing decision can become a high hidden cost over time.
3-2 Regulatory Control and Process Validation
Under ISO13485 requirements, each production batch must include comprehensive process and inspection records. These records encompass machining parameters, tooling logs, in-process data, and final inspection results. During first article validation, teams typically conduct full-dimensional inspections to verify critical features and geometric relationships.
When production volumes are very low, the full documentation and inspection requirements still apply, even if only a small number of parts are produced. For the subsequent batch, records must be reopened, parameters reviewed, and approvals repeated. The compliance workload does not decrease in proportion to the quantity produced.
In this context, MOQ represents the minimum quantity necessary to justify the validation and documentation efforts required for regulated medical device production.
3-3 Statistical Validation of Process Capability
Before full production release, manufacturers conduct process validation using defined sample sizes to confirm repeatability and stability. Metrics such as Cpk evaluate how critical dimensions perform across consecutive cycles, ensuring that variation remains within a predictable range.
This stage demands focused engineering efforts and precise metrology. When sample sizes are too small, capability results may appear acceptable but can mask the true process behavior. Early data may seem stable initially, then shift as volume increases and more parts are measured.
When that occurs, teams must retune parameters and repeat capability studies, which delays scale-up. Establishing a realistic MOQ provides sufficient volume to generate meaningful validation data early on, thereby reducing the risk of instability during ramp-up.
4. How Annual Volume Forecasts Influence MOQ Flexibility
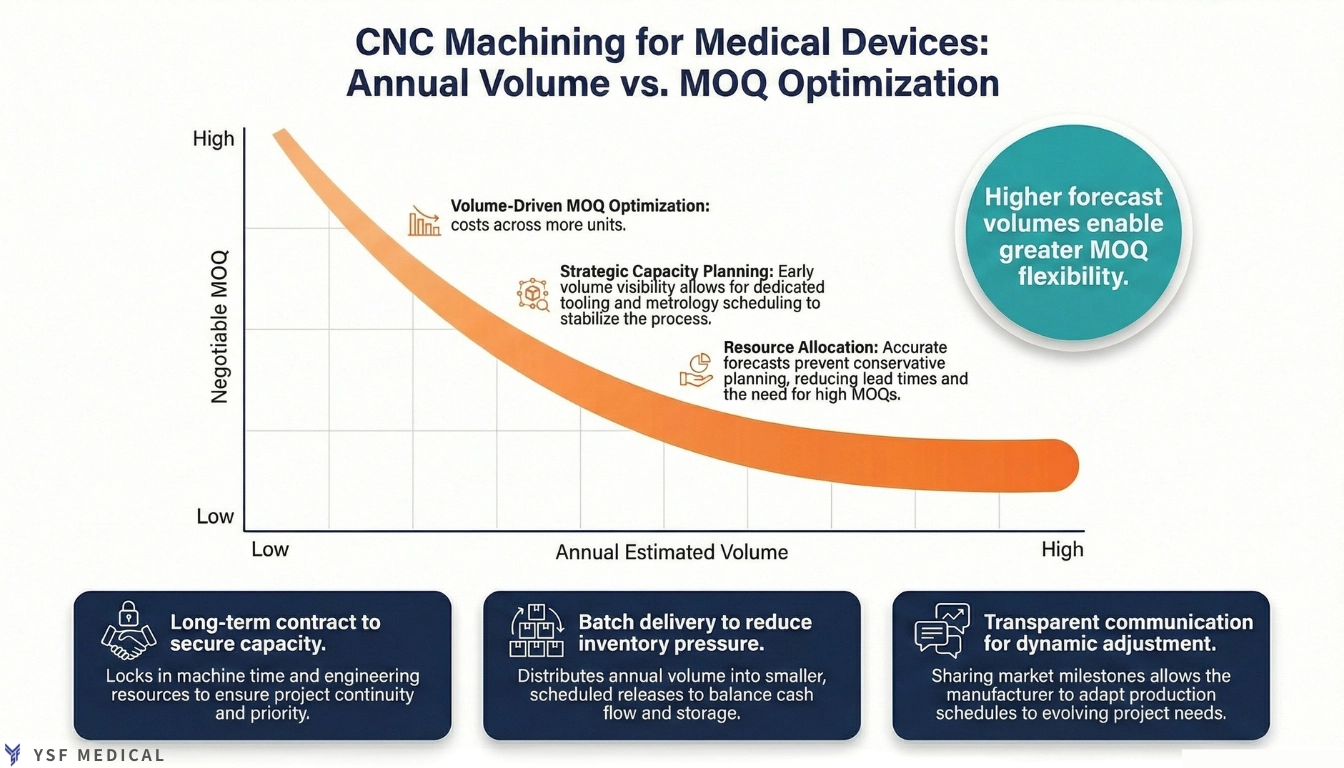
4-1 Capacity Planning and Long-Term Order Strategy
Providing an estimated annual volume range early enables manufacturers to plan capacity, procure materials, and allocate workforce effectively. It also facilitates decisions regarding dedicated fixtures, project tooling, and metrology scheduling.
When forecasts are uncertain, planning tends to be conservative. Tooling investments may be postponed, and capacity remains limited. If demand increases suddenly, resources cannot scale rapidly, resulting in longer lead times and reduced flexibility in MOQ.
For programs with steady demand, production can be organized into scheduled batches that distribute setup and fixture costs across consistent volumes. Long-term collaboration extends beyond pricing and often determines whether launch and clinical timelines remain on track.
4-2 Transparency Enhances Operational Flexibility
In the early stages of development, exact volumes may be uncertain. However, sharing the expected market scope, trial timelines, and launch targets provides manufacturers with sufficient context to plan capacity, manage changeover risks, and schedule fixtures, tooling, and metrology in the correct sequence.
Without this visibility, projects enter the standard production queue with limited priority planning. When pilot builds or scale-up phases begin, machine and inspection constraints emerge. Reallocating resources requires time, often leading to stricter MOQ requirements and longer lead times than initially quoted.
For sourcing and engineering teams, transparency transforms the MOQ into a dynamic planning tool that adapts as the program progresses. Without this transparency, MOQ becomes a late-stage constraint that intensifies pressure when timelines are already tight.
5-1 How Part Design Influences MOQ Thresholds
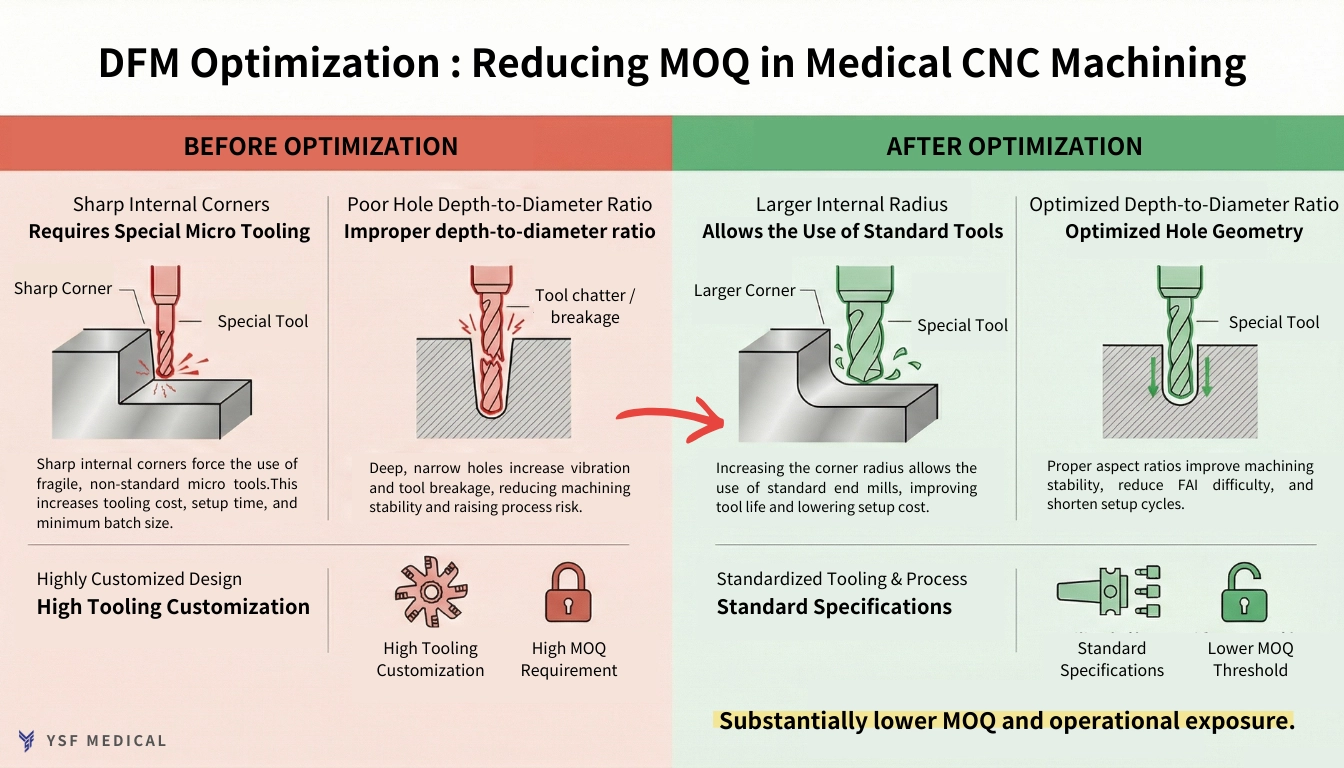
DFM(Design for Manufacturability) review, issues frequently arise during pilot builds. Limitations in tool reach, the need for custom cutters, and additional secondary operations increase setup time and extend first article validation. Consequently, each subsequent batch requires the same level of preparation, effectively raising the practical MOQ.
When designs align with existing machining platforms and measurement methods, parameter transfer becomes more seamless, and validation time decreases. As the process stabilizes, MOQ flexibility improves.
5-2 Bridging Development and Production
Many programs begin with prototypes from one supplier and later transition to another for production. Once the transfer occurs, the production site must repeat validation, first article inspection, and quality documentation. In regulated medical programs, each transition necessitates rebuilding audit-ready records from scratch.
When prototypes are developed without considering DFM, machining conditions, and fixturing rarely transfer seamlessly to production. As a result, the production team must readjust parameters and reestablish process capability, which slows progress and compresses launch timelines.
When a production-capable partner is involved early, and prototypes are tested under real manufacturing conditions, validated parameters and inspection methods can transition directly into scale-up. This continuity reduces redundant work and facilitates a smoother product introduction.
5-3 The Advantages of a Vertically Integrated Supply Chain
In a multi-supplier model, milling, heat treatment, and surface finishing are managed by different companies, each operating under its own batch rules and schedules. The minimum batch size at any stage can constrain the entire program, effectively raising the MOQ beyond initial expectations.
When parts move between facilities, they must be packed, transferred, received, and re-inspected. Delays at any stage impact subsequent steps, and each handoff increases the risk of dimensional or surface variations.
When machining, heat treatment, and finishing operate under a unified quality system, batch planning and documentation remain aligned. This integration promotes a smoother process flow, reduces MOQ pressures, and simplifies the management of supply chain risks.
Q1:Why do MOQ quotes vary so widely for the same part?
MOQ differences typically reflect the organizational structure of each manufacturer. Larger facilities with multiple similar machines can distribute setup and changeover times across more jobs, enabling lower minimum order quantities. In contrast, smaller shops with limited capacity often require higher batch sizes to maintain efficient utilization.
The key question is not the MOQ itself, but whether the supplier has produced similar components under comparable conditions and can provide stable batch records and inspection documentation. While a lower MOQ may seem appealing in the quote, repeated variations and adjustments can ultimately extend the overall timeline.
Q2:Our product is still in development, and the volume is uncertain. How should we approach discussions about MOQ?
In the early stages of development, discussions should not focus solely on exact quantities. Sharing information about design maturity, regulatory timelines, and market milestones provides manufacturers with sufficient context to plan capacity, assess changeover risks, and allocate resources more effectively.
A common strategy is to accept a higher unit price in exchange for a lower initial MOQ. This approach limits early inventory exposure while maintaining flexibility. Once the design stabilizes and demand becomes clearer, batch structure and pricing can be adjusted under more predictable production conditions.
If you are evaluating your MOQ strategy during development, please feel free to contact us at sales@ysfbone.com. We can review your program stage and provide technical feedback based on actual production conditions.
Q3:Why does MOQ differ significantly between the prototype and production phases?
During the prototype phase, the primary goal is to confirm manufacturability. Engineering efforts focus on testing machining conditions, refining parameters, and verifying feasibility. At this stage, technical considerations take precedence over part quantity.
Once the program transitions into production, priorities shift to batch stability, heat traceability, and comprehensive documentation control. These requirements apply regardless of order size. The production MOQ reflects the effort required to maintain a controlled and audit-ready environment.
Q4:Can similar parts be combined to meet MOQ requirements?
Combining parts requires more than just dimensional similarity. The key consideration is whether they can share the same material batch, machining strategy, and fixturing setup.
If the material grade, tolerance level, or surface treatment differs, each part typically requires separate validation and inspection. Even when drawings appear similar, traceability and compliance controls often restrict the advantages of merging batches. In many cases, any time savings are negated once documentation and quality checks are implemented.
If you would like to explore whether components can be consolidated for batch planning, please contact us at sales@ysfbone.com. We can evaluate process compatibility and assess the impact on compliance before implementing any changes.
Q5:Unit costs are high at low volumes, but large batches increase inventory risk. Is there a balanced approach?
A balanced approach involves aligning on estimated annual demand and scheduling deliveries in planned releases. This strategy keeps inventory risk manageable while providing the manufacturer with sufficient visibility to plan capacity, materials, and inspection resources effectively.
It is also helpful to review the cost structure collaboratively. When material costs, machining time, setup efforts, and quality workloads are clearly understood, teams can identify which factors are volume-driven and which pertain to process design. This clarity supports a delivery rhythm that balances cash flow with production stability.
For teams considering staggered releases or long-term supply planning, contact us at sales@ysfbone.com to initiate a structured discussion on sustainable batch strategies.
In medical device CNC machining, MOQ refers to the minimum quantity required to support setup validation, fixturing, metrology, documentation, and process stability. When this foundational volume is underestimated, risks do not disappear; instead, they manifest later as yield variation, extended lead times, or repeated validation cycles.
Comparing MOQ figures alone rarely provides the full picture. A more comprehensive evaluation considers whether the supplier can maintain process capability at actual production volumes, whether scalable fixturing and measurement systems are implemented, and whether pilot-stage parameters transition smoothly into full-scale production.
Stability improves when DFM and machining feasibility are addressed early, and when sourcing provides realistic volume expectations and milestones. In this environment, the MOQ evolves with product maturity rather than becoming a last-minute constraint. A mature strategy balances process stability, validation workload, and supply cadence, resulting in more predictable ramp-up and delivery performance.
YSF Medical specializes in precision machining for medical devices, supporting programs from early design through pilot validation to full production. Operating under an ISO 13485 quality management system, we ensure traceability, document control, and process stability from project initiation.
We operate advanced CNC milling and mill-turn platforms, choosing the approach based on geometry and tolerance requirements. Prototypes are produced under production-ready conditions, allowing pilot data to transition seamlessly into volume manufacturing.
Our team actively participates in DFM discussions to align design specifications, tooling accessibility, and critical tolerances with stable machining practices. We support staged deliveries and implement structured capacity planning to balance inventory risk while ensuring long-term process consistency.
For MOQ planning or pilot-to-production transition support, please contact sales@ysfbone.com to discuss your program requirements.
This article is provided for informational purposes only and is intended for professionals in the medical device industry. It does not constitute engineering, regulatory, or legal advice.Readers are responsible for evaluating the applicability of this information to their specific products and regulatory jurisdictions. Any technical references, including dimensions, parameters, or timelines, are illustrative only and may vary in practice.
While reasonable efforts have been made to ensure accuracy, no representations or warranties are made, and the authors and publisher disclaim any liability for losses arising from reliance on this content.All case examples have been anonymized to protect confidentiality.
10. References
TFDA
https://www.fda.gov.tw/
ISO 13485
https://www.iso.org/standard/59752.html
ISO 9001:2015 Quality management systems — Requirements
https://www.iso.org/standard/62085.html
ISO 10993-1:2025 Biological evaluation of medical devices
https://www.iso.org/standard/10993-1
FDA Medical Devices
https://www.fda.gov/medical-devices
Standard Specification for Polyetheretherketone (PEEK) Polymers for Surgical Implant Applications
https://store.astm.org/f2026-17.html
FDA charges a fee for review of Premarket Notifications [510(k)]
https://www.fda.gov/medical-devices/premarket-submissions-selecting-and-preparing-correct-submission/premarket-notification-510k
Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC (Text with EEA relevance. )
https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32017R0745
ASM International – Materials Information and Engineering Resources
https://www.asminternational.org/
https://www.nist.gov/manufacturing
NIST – Manufacturing Extension Partnership & Precision Measurement Standards
© 2026 YSF Medical Enterprise Co., Ltd. | Experts in Orthopedic Implants and Surgical Instruments Manufacturing | ISO 13485-Certified Medical Device Manufacturer | Precision CNC Machining | All rights reserved. Please cite the source when quoting or reproducing any content.
.webp) Contact Us
Contact Us


.webp) CONTACT US
CONTACT US

